Fluorophores for biological applications
Fluorophores are molecules (for biological applications usually polyaromatic hydrocarbons or heterocycles) with specific fluorescent properties, such as characteristic excitation or emission spectra. They can be used as fluorescent probes if they are targetted to specific locations within cell specimens (see for more information).
Excitation of fluorophores
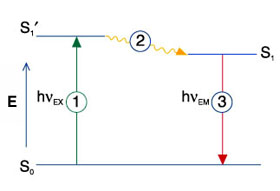
Fluorophores can be excited by irradiating them with photons of sufficient energy (filtered light, lasers). When the fluorophore is excited, a singlet state of the molecule with a higher energy is generated (see Jablonski diagram above S1'). This excited state is in the range of 1-10 ns and this fluorescence lifetime is characteristic for a specific fluorophore in a given environment and thus provides an additional parameter that can be measured (see FLIM). The energy of the excited singlet state molecule partially dissipates in heat (see 2) leading to a relaxed single state (see S1). Each fluorophore has a characteristic excitation spectrum, which helps to find the right excitation light (e.g. laser line) for microscopic detection (see Fluorescence Spectra Viewer).
Fluorescence emission
When the fluorophore returns from its excited state to the ground state (S0), it emits a photon with a lower energy compared to the exciting photon. The difference between the wavelengths of excitation and emission photons is called the Stoke's shift and is caused by the heat dissipation during the time in the excited state. For detection of emitted light with light microscopes a large Stoke's shift is desirable because it facilitates the separation of excitation light from emitted light. Fluorescence is a cyclic process that allows the same fluorophore running through many cycles of excitation and photon emission until it gets irreversibly destroyed by a process called photobleaching.
Fluorescence spectra
For the design of a microscopic experiment it is essential to know the excitation and emission spectra of the fluorophores to be used. This allows the right choice of laser lines for excitation and filters and detectors for the recording of the emitted light. When several fluorophores are sued in the same specimen, ideally one laser line should only excite asingle fluorophore. Accordingly, the emission spectra should be well separated to guarantee the detection of only one fluorophore emission at a time. This is to avoid cross-talk between spectrally different channels, which would lead to false imaging results. To achieve this, the fluorophores should be chosen to optimally match the technical components such as lasers, dichroics, emission filters, mirrors and detectors.
An update of this page will follow in due course